BEPANTHENE BAYER 250 mg/ml, solution for injection I.M. 6 ampoules of 2ml
Indications: Adjunctive treatment of diffuse alopecia.
Bepanthene 250 mg/ml is used in case of diffuse hair loss. A box contains 6 ampoules of 2 m.
Free delivery for orders over 89€*.
* in metropolitan France and excluding drugs

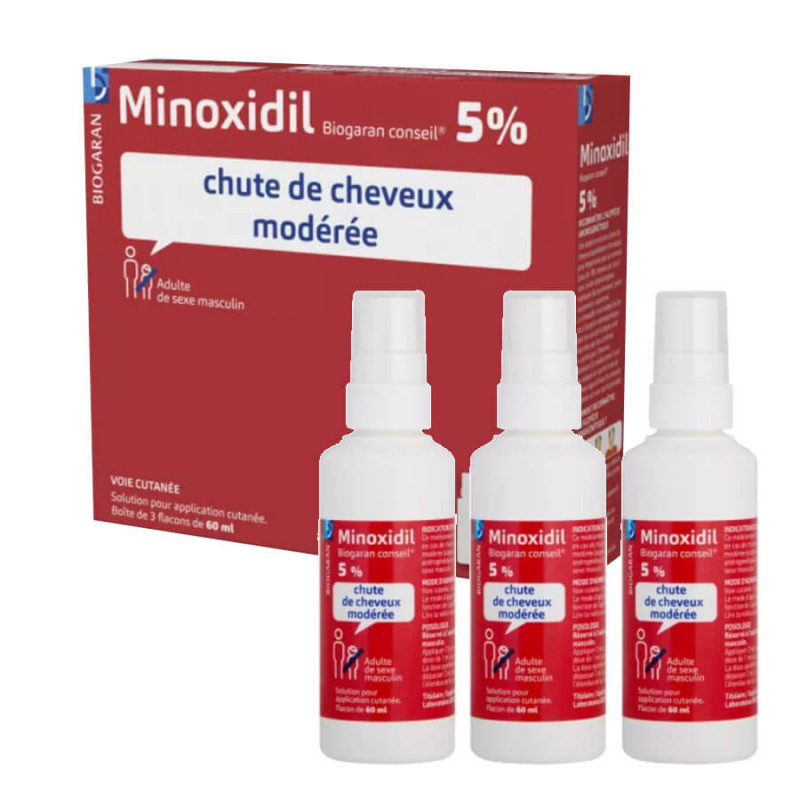
This medication is indicated for moderate hair loss (androgenetic alopecia) in males.
This medicine is indicated for moderate hair loss (androgenetic alopecia) in males.
Directions for use
For adult males only.
Apply twice a day (once in the morning and once in the evening) a dose of 1 ml to the bald area of the scalp, starting from the center of the area to be treated. This dose must be respected regardless of the size of the area concerned.
The total daily dose should not exceed 2 ml.
Mode of administration
Cutaneous way.
Spread the product with the fingertips so as to cover the entire area to be treated.
After applying the solution, wash your hands thoroughly.
Do not apply the product to any other part of the body.
Apply to thoroughly dry hair and scalp.
The method of application varies according to the delivery system used with the bottle.
Sprayer: This system is suitable for application to large areas.
Remove the small transparent cap and direct the sprayer towards the center of the surface to be treated, press once and spread the product with the fingertips. Repeat 6 times to apply a 1 ml dose.
Avoid inhaling the product.
After application, remove the sprayer, rinse it and screw the child-resistant cap back on the bottle. Sprayer with applicator: this system is suitable for application on small surfaces or under the hair.
1. Remove the small clear cap and pull up firmly to remove the "head" of the sprayer.2. Insert the applicator onto the sprayer stem (now without the head) and push it in fully.
3. Point the sprayer at the center of the area to be treated or under the hair, squeeze once and spread the product with your fingertips. Repeat 6 times to apply a 1 ml dose.
Avoid inhaling the product.
After application, remove the applicator and sprayer, rinse them and screw the child-resistant cap back on the bottle.
Duration of use
Results may not be seen for at least 2 to 4 months of twice-daily treatment. The onset and degree of response will vary with each individual.
Do not discontinue applications as this may result in a return to the previous condition after 3 to 4 months.
Do not use minoxidil if there is no improvement after 4 months of application.
Precautions for use
Contraindications:
- Hypersensitivity to the active substance or to any of the excipients mentioned in the section List of excipients.
- Scalp diseases.
- Poor tolerance to the 2% form, whatever the symptomatology.
- This drug is contraindicated in women.
- In subjects under 18 years of age or over 65 years of age.
Warnings and other special precautions:
Topical minoxidil should be used on a normal, healthy scalp and should not be administered if the scalp skin is inflamed, infected, irritated or painful.
Increased percutaneous absorption of minoxidil, which may cause systemic effects, may occur in the presence of :
- dermatosis or lesion of the scalp,
- concomitant application of retinoic acid, anthralin, or other irritating topical agents
- increase of the applied dose and/or increase of the frequency of the applications: it is imperative to respect the dosage and the mode of administration.
Similarly, although extensive use of minoxidil solution has not revealed any systemic effects, it cannot be excluded that increased absorption due to individual variability or unusual sensitivity may cause systemic effects. Patients should be advised of this.
Patients should be monitored regularly for symptoms suggestive of systemic effects, such as decreased blood pressure, tachycardia, chest pain, fainting, dizziness, unexplained weight gain, or signs of fluid retention (swelling of the hands or feet) as a precautionary measure.
Monitoring should be done at the beginning of treatment and then on a regular basis. If severe systemic or dermatologic reactions or any other unexpected symptoms occur (see Adverse Reactions), treatment should be discontinued and the patient should consult a physician.
Patients with cardiovascular disease or cardiac arrhythmia should consult a physician before using topical minoxidil. In these patients, the benefit of treatment should be weighed. In particular, they should be warned of potential adverse effects so that treatment can be discontinued if any of these occur and a physician notified.
If symptoms persist or worsen, or if new symptoms develop, patients should discontinue treatment and consult a physician.
Do not apply minoxidil:
- if hair loss occurs in patches and/or abruptly, if hair loss occurs as a result of disease or medication, or if the reason for the alopecia is unknown.
- on another part of the body.
- at the same time as other medications on the scalp
In case of accidental contact with the eye, an irritated skin, a mucous membrane, the solution can cause a burning sensation and/or an irritation. In case of contact with sensitive surfaces (eye, irritated skin, mucous membranes), rinse thoroughly with cold running water.
Accidental ingestion may result in severe adverse effects, particularly cardiac (see Adverse Reactions). This product should therefore be kept out of the reach of children.
Sun exposure is not recommended when using minoxidil.
Some patients have experienced changes in hair color and/or texture.
Do not inhale.
Use of a higher dosage or frequency will not improve results.
Continued treatment is necessary to increase and maintain hair growth. Otherwise, hair loss will resume.
A temporary increase in hair loss may occur during the first 2 to 6 weeks of treatment. If hair loss persists, the patient should stop using MINOXIDIL BIOGARAN CONSEIL and consult their doctor.
Unwanted hair growth may occur due to transfer to areas other than the scalp
This medicine contains 518.5 mg of propylene glycol per 1 ml dose. It may cause skin irritation.
This medication contains 250 mg of alcohol (ethanol) per 1ml dose. It may cause a burning sensation on damaged skin. Keep away from any flame, lighted cigarette or source of intense heat (e.g. hair dryer).
Pregnancy and breastfeeding:
Pregnancy
This medication is contraindicated in women.
Breastfeeding
When administered systemically, minoxidil passes into breast milk.
This drug is contraindicated in women.
composition
solution for cutaneous application (clear, slightly yellow to pink with a form of alcohol odour): Minoxidil 5 g/100 ml.
Excipients: Propylene glycol, ethanol 96%, purified water.
Notable Excipients:
Each ml of solution contains 518.5 mg of propylene glycol and 250 mg of 96% ethanol.
Indications: Adjunctive treatment of diffuse alopecia.
Bepanthene 250 mg/ml is used in case of diffuse hair loss. A box contains 6 ampoules of 2 m.
Indications: This medicine is indicated in case of moderate hair loss (androgenetic alopecia) in adults, men or women. It promotes hair growth and stabilizes the phenomenon of hair loss.
Description: This medication is indicated for moderate hair loss (androgenetic alopecia) in males.
Description: This medication contains minoxidil, a substance that stimulates hair growth and slows hair loss. Minoxidil is also a vasodilator used orally in the treatment of high blood pressure. When applied topically to the scalp, the antihypertensive effect of minoxidil is usually unnoticeable.
Indications: This medicine is recommended in the adjunctive treatment of disorders of corneal healing and in cases of fragile nails or hair.
Bepanthene 100 mg 60 tablets is a medication used as an adjunct treatment for people suffering from hair loss (diffuse alopecia).
Description: This medication contains minoxidil, a substance that stimulates hair growth and slows hair loss when applied to the scalp. Minoxidil is also a vasodilator used orally in the treatment of high blood pressure. When applied topically to the scalp, the anti-hypertensive effect of minoxidil is usually unnoticeable.
This medication contains minoxidil, a substance that stimulates hair growth and slows hair loss when applied to the scalp.
Indications: This drug is indicated in cases of moderate hair loss (androgenetic alopecia) in males.
It is not recommended for women because of the high frequency of hypertrichosis (abnormal development of the hair system) at a distance from the application sites.
This medicine is indicated in case of moderate hair loss (androgenic alopecia) in adults, men or women. It promotes hair growth and stabilizes the phenomenon of hair loss.
Indications : Treatment of hair loss. Solution for cutaneous application.
3 x 60ml bottles with pipette
This medication is indicated for moderate hair loss (androgenic alopecia) in males.
Indications: This medicine is recommended in the adjunctive treatment of disorders of corneal healing and in cases of fragile nails or hair.
Minoxidil Cooper 5 Percent is a medicine containing minoxidil, a substance that stimulates hair and beard growth and slows hair loss when applied to the scalp.
This Anti-Hair Loss and Growth serum has been specifically tested as a companion product to the leading topical drug treatments on areas harvested and implanted for hair transplantation. It can be used alone or in combination with a local drug treatment.
Application on dry or wet scalp
8 sprays / Daily use
Indications: This medicine is recommended in the adjunctive treatment of disorders of corneal healing and in cases of fragile nails or hair.