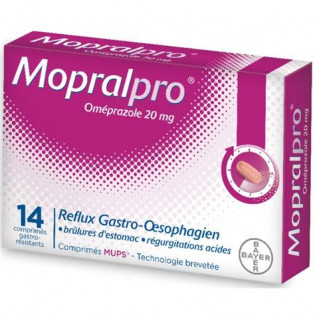
Description Viatris Esomeprazole 20mg 14 capsules heartburn
Gastro-resistant capsule. Capsule with opaque yellow head and opaque white body with "20 mg" printed in black on head and body.
The capsule contains off-white to grayish spherical microgranules.
INDICATIONS Viatris Esomeprazole 20mg 14 capsules heartburn
ESOMEPRAZOLE MYLAN CONSEIL is indicated for the short-term treatment of symptoms of gastroesophageal reflux disease (e.g. pyrosis and acid regurgitation) in adults.
DOSAGE AND ADMINISTRATION Viatris Esomeprazole 20mg 14 capsules
Dosage
The recommended dose is 20 mg of esomeprazole (one capsule) per day. It may be necessary to take the capsules for 2 or 3 consecutive days to obtain an improvement in symptoms.
Treatment may last up to 2 weeks.
Once symptoms have disappeared, treatment should be discontinued. If symptoms persist after 2 weeks of continuous treatment, the patient should be advised to consult a doctor.
Special populations Patients with renal impairment
No dosage adjustment is necessary in patients with renal impairment.
Patients with severe renal impairment should be treated with caution due to limited experience in these patients (see Pharmacokinetics). Patients with hepatic impairment
No dosage adjustment is necessary in patients with mild to moderate hepatic impairment.
However, patients with severe hepatic impairment should be advised by a physician before taking ESOMEPRAZOLE MYLAN CONSEIL (see Pharmacokinetics).
Elderly (> 65 years) No dosage adjustment is necessary in the elderly.
Pediatric population There is no justified use of ESOMEPRAZOLE MYLAN CONSEIL in the pediatric population under 18 years of age for the indication: "short-term treatment of symptoms of gastroesophageal reflux disease (e.g. pyrosis and acid regurgitation)".
Method of administration
Capsules should be swallowed whole with half a glass of water. Capsules should not be chewed or crunched.
For patients with swallowing difficulties: Capsules may also be opened and the contents dispersed in half a glass of non-carbonated water.
No other liquid should be used, as the enteric coating may be dissolved.
Stir and drink the liquid with the granules immediately or within 30 minutes. Rinse glass with half a glass of water and drink.
Gastro-resistant granules must not be chewed.
CONTRAINDICATIONS Viatris Esomeprazole 20mg 14 capsules
Known hypersensitivity to esomeprazole, benzimidazole derivatives or any of the excipients (see Composition). Esomeprazole should not be given concomitantly with nelfinavir (see Interactions).
WARNINGS and PRECAUTIONS
General Patients are advised to seek medical advice in the event of : Significant and unintentional weight loss, repeated vomiting, dysphagia, hematemesis or melena, and if gastric ulcer is suspected or present, the possibility of malignancy should be excluded, as treatment with esomeprazole may alleviate symptoms and delay diagnosis.
History of gastric ulcer or digestive surgery. Continuous symptomatic treatment for indigestion or heartburn for 4 weeks or more.
Jaundice or severe liver disease. New onset or recent modification of symptoms in patients over 55 years of age. Patients suffering from persistent and recurrent disorders of the difficult digestion (dyspepsia) or heartburn (pyrosis) type should consult their doctor regularly.
Patients over 55 years of age taking non-prescription daily medication for difficult digestion or heartburn should inform their doctor or pharmacist.
Patients should not take Esomeprazole Mylan Conseil as a long-term preventive medication. Treatment with proton pump inhibitors (PPIs) may lead to a slight increase in the risk of gastrointestinal infections, including Salmonella and Campylobacter, and possibly Clostridium difficile in hospitalized patients (see Pharmacodynamics).
Patients should consult their doctor before taking this drug if endoscopy or urea breath testing is planned. The combination of esomeprazole with atazanavir is not recommended (see Pharmacodynamics).
If the combination of atazanavir with a proton pump inhibitor is deemed essential, close clinical monitoring is recommended, together with an increase in the dose of atazanavir to 400 mg with 100 mg ritonavir. A dose of 20 mg of esomeprazole should not be exceeded.
Esomeprazole is a CYP2C19 inhibitor. At the start or end of treatment with esomeprazole, the risk of interactions with drugs metabolized by CYP2C19 should be considered.
An interaction between clopidogrel and esomeprazole has been observed. The clinical relevance of this interaction is uncertain.
Concomitant use of esomeprazole and clopidogrel should be avoided (see Interactions). Patients should not take other PPIs or anti-H2 drugs concomitantly.
Interference with laboratory tests Increased levels of Chromogranin A (CgA) may interfere with tests performed to investigate neuroendocrine tumours.
To avoid such interference, esomeprazole treatment should be discontinued at least 5 days before CgA levels are measured (see Pharmacodynamics). If CgA and gastrin levels are not normalized after the initial measurement, measurements should be repeated 14 days after discontinuation of the proton pump inhibitor.
Subacute cutaneous lupus erythematosus (SCLE) Proton pump inhibitors are associated with very occasional cases of SCLE. If lesions develop, particularly on sun-exposed skin, and are accompanied by arthralgia, the patient should seek prompt medical attention, and the healthcare professional should consider discontinuing esomeprazole. The occurrence of LECS after treatment with a proton pump inhibitor may increase the risk of LECS with other proton pump inhibitors. Sucrose This medicine contains sucrose spheres.
Patients with fructose intolerance, glucose-galactose malabsorption syndrome or sucrase/isomaltase deficiency (rare hereditary diseases) should not take this medicine. Parahydroxybenzoates This medicine contains parahydroxybenzoates and may cause allergic reactions (possibly delayed). Sodium benzoate This drug contains 0.0018 mg sodium benzoate in each 20 mg capsule. Sodium This drug contains less than 1 mmol sodium per capsule (23 mg), i.e. it is "essentially sodium-free".
INTERACTIONS Viatris Esomeprazole 20mg 14 capsules
Drug interactions: Interaction studies were performed in adults only.
Effects of esomeprazole on the pharmacokinetics of other drugs As esomeprazole is an enantiomer of omeprazole, reported interactions with omeprazole should be taken into consideration.
Protease inhibitors: Omeprazole has been reported to interact with certain protease inhibitors. The clinical significance and mechanisms of these interactions are not always known. Increased gastric pH during treatment with omeprazole may alter the absorption of protease inhibitors.
Other possible interaction mechanisms involve CYP2C19 inhibition.
For atazanavir and nelfinavir, a decrease in plasma concentrations of these drugs has been reported when administered concomitantly with omeprazole; concomitant administration of omeprazole and these drugs is therefore not recommended. Concomitant administration of omeprazole (40 mg once-daily) with atazanavir 300 mg/ritonavir 100 mg in healthy volunteers resulted in a substantial decrease in atazanavir plasma concentrations (approximately 75% decrease in AUC, Cmax and Cmin).
Increasing the dosage of atazanavir to 400 mg did not compensate for the effect of omeprazole on atazanavir plasma concentrations. Concomitant administration of omeprazole (20 mg once daily) and atazanavir 400 mg/ritonavir 100 mg in healthy volunteers resulted in an approximately 30% decrease in atazanavir exposure, compared with the exposure observed after administration of atazanavir 300 mg/ritonavir 100 mg once daily, without omeprazole 20 mg once daily.
Concomitant administration of omeprazole (40 mg once daily) decreased the mean AUC, Cmax and Cmin of nelfinavir by 36-39% and the mean AUC, Cmax and Cmin of its pharmacologically active metabolite M8 by 75-92%.
FERTILITY/PROGNANCY/LACTATION Viatris Esomeprazole 20mg 14 capsules
Pregnancy
A moderate amount of data in pregnant women (between 300-1000 pregnancy outcomes) has not demonstrated any malformative or toxic effects of esomeprazole on the fetus or the newborn.
Animal studies have shown no direct or indirect deleterious effects on reproduction (see Preclinical safety).
As a precautionary measure, ESOMEPRAZOLE MYLAN CONSEIL should not be used during pregnancy.
Breast-feeding
It is not known whether esomeprazole/metabolites are excreted in breast milk.
There are no adequate data on the effects of esomeprazole in neonates/infants.
Esomeprazole should not be used during breast-feeding. Fertility Animal studies with racemic omeprazole, administered orally, show no effect on fertility.
Operation and use of machinery
Esomeprazole has a minor influence on the ability to drive and use machines.
Undesirable effects such as dizziness and visual disturbances are uncommon (see Undesirable effects).
Patients experiencing such adverse reactions should not drive or operate machinery.
ADVERSE REACTIONS Viatris Esomeprazole 20mg 14 capsules
Headache, abdominal pain, diarrhea and nausea are among the adverse reactions most frequently reported in clinical trials (and also in post-marketing use).
Moreover, the safety profile is similar across formulations, treatment indications, age groups and patient populations. No dose-related adverse effects have been identified.
OVERDOSAGE (top of page) To date, experience of deliberate overdosage is very limited.
Symptoms described after 280 mg are gastrointestinal symptoms and signs of fatigue. Single daily doses of 80 mg have been well tolerated.
No specific antidote is known. Esomeprazole is strongly bound to plasma proteins and is therefore not readily dialyzable. In the event of overdosage, appropriate symptomatic treatment should be instituted.
PHARMACODYNAMICS
Pharmacotherapeutic class: drugs for acidity disorders, proton pump inhibitors; ATC code: A02BC05.
Esomeprazole is the S-isomer of omeprazole and reduces gastric acid secretion by a specifically targeted mechanism of action.
It is a specific inhibitor of the proton pump at the parietal cell level. The two R and S isomers of omeprazole have similar pharmacodynamic activity. Mechanism of action Esomeprazole is a weak base.
It is concentrated and converted to its active form in the acidic environment of the secretory canaliculi of the parietal cell, where it inhibits the enzyme H+K+-ATPase (the proton pump), basal acid secretion and stimulated acid secretion. Pharmacodynamic effects After oral doses of 20 and 40 mg of esomeprazole, the effect occurs within one hour.
After repeated administrations of 20 mg of esomeprazole once a day for 5 days, the mean acid peak obtained after pentagastrin stimulation fell by 90% on the 5th day, 6 to 7 hours after dosing. After 5 days of oral doses of 20 mg and 40 mg esomeprazole, intragastric pH above 4 was maintained for an average of 13 hours and 17 hours over 24 hours respectively in patients with symptomatic gastro-oesophageal reflux disease (GERD).
The percentages of patients whose intragastric pH remained above 4 for at least 8, 12 and 16 hours after taking 20 mg of esomeprazole were 76%, 54% and 24% respectively. With a 40 mg dose, the corresponding percentages were 97%, 92% and 56%. Using the area under the curve (AUC), as a parameter reflecting plasma concentration, a relationship between inhibition of acid secretion and exposure was demonstrated. During treatment with antisecretory drugs, serum gastrin concentration increases in response to reduced acid secretion.
Similarly, CgA levels rise in response to reduced gastric acidity. Increased CgA levels may interfere with tests performed to investigate neuroendocrine tumors. According to published data, proton pump inhibitors should be discontinued between 5 days and 2 weeks before measuring CgA levels.
The aim is to allow CgA levels artificially increased by PPIs to return to normal. An increase in the number of ECL cells, possibly related to the rise in serum gastrin levels, has been observed in some patients treated long-term with esomeprazole. Decreased gastric acid secretion, whatever the cause, particularly that induced by proton pump inhibitors (PPIs), increases the number of bacteria normally found in the gastrointestinal tract.
Treatment with PPIs may slightly increase the risk of gastrointestinal infections caused by germs such as Salmonella and Campylobacter, and possibly Clostridium difficile, in hospitalized patients.
Clinical efficacy
Esomeprazole 20 mg has been shown to effectively treat frequent heartburn in subjects receiving a dose every 24 hours for 2 weeks.
In two multicenter, randomized, double-blind, placebo-controlled pivotal studies, 234 subjects with a recent history of frequent heartburn were treated with 20 mg of esomeprazole for 4 weeks. Symptoms associated with acid reflux (such as heartburn and acid regurgitation) were assessed retrospectively over a 24-hour period.
In both studies, esomeprazole 20 mg was significantly more effective than placebo on the primary endpoint, complete resolution of heartburn defined as the absence of heartburn in the 7 days prior to the final visit (33.9% - 41.6% vs. 11.9% - 13.7% for placebo (p< 0.001). The secondary endpoint, complete resolution of heartburn defined as no heartburn recorded in the patient diary for 7 consecutive days, was statistically significant at both week 1 (10.0% - 15.2% vs. 0.9% - 2.4% on placebo, p= 0.014, p< 0.001) and week 2 (25.2% - 35.7% vs. 3.4% - 9.0% on placebo, p< 0.001).
The time to sustained resolution of heartburn, defined as 7 consecutive days without heartburn since the first heartburn episode, was significantly shorter in the 20 mg esomeprazole group (39.7% - 48.7% at day 14 vs. 11.0% - 20.2% on placebo).
The median time to first resolution of nocturnal heartburn was 1 day, statistically significant versus placebo in one study (p = 0.048) and close to significance in the other (p = 0.069). Around 80% of nights were heartburn-free in all periods, and 90% of nights were heartburn-free in the second week of each trial, compared with 72.4% to 78.3% for placebo.
PRECLINICAL SAFETY
Non-clinical data from conventional studies of safety pharmacology, repeated-dose toxicology, genotoxicity, carcinogenesis, and reproductive and developmental functions did not reveal any particular risk for humans.
The following adverse effects have not been observed in clinical studies, but have been observed in animals subjected to exposure levels similar to those used for humans and could be of clinical significance.
Carcinogenesis studies in rats with the racemic mixture showed gastric ECL cell hyperplasia and carcinoid tumors.
In rats, these gastric changes are the result of prolonged and significant hypergastrinemia, secondary to reduced gastric acid secretion, and are observed in this animal during long-term treatment with gastric acid secretion inhibitors.
SHELF LIFE
2 years For the bottle: After first opening, store for a maximum of 3 months.
SPECIAL STORAGE PRECAUTIONS
Store at temperatures not exceeding 25°C.
Bottle: Keep bottle tightly closed, away from moisture. Blisters: Store in original outer packaging, protected from moisture.
Composition Viatris Esomeprazole 20mg 14 capsules heartburn
Each capsule contains 20 mg esomeprazole (as esomeprazole magnesium dihydrate).
Notable excipient(s): each capsule contains 8.05 mg sucrose, 1.85 micrograms methyl parahydroxybenzoate (E218), 0.56 micrograms propyl parahydroxybenzoate (E216) and 1.8 micrograms sodium benzoate (E211).
Excipients: Capsule contents : Sugar spheres (sucrose, corn starch), hypromellose, diméticone emulsion 35% (diméticone, propyl parahydroxybenzoate (E216), methyl parahydroxybenzoate (E218), sorbic acid, sodium benzoate, polyethylene glycol sorbitan monolaurate, octylphenoxy-polyethoxy-ethanol and propylene glycol), polysorbate 80, mannitol, diacetylated monoglycerides, talc, 30% methacrylic acid/ethyl acrylate copolymer dispersion (1 : 1) (copolymer of methacrylic acid and ethyl acrylate, sodium laurilsulfate and polysorbate 80), triethyl citrate, stearic macroglycerides Capsule shell: Black iron oxide (E172), shellac, yellow iron oxide (E172), titanium dioxide (E171), gelatin.
 Français
Français English
English