SINOVIAL 3 PREFILLED SYRINGES OF 2ML
Sinovial® is indicated in cases of pain or reduced mobility in order to restore the physiological and rheological properties of arthritic joints.
Free delivery for orders over 89€*.
* in metropolitan France and excluding drugs

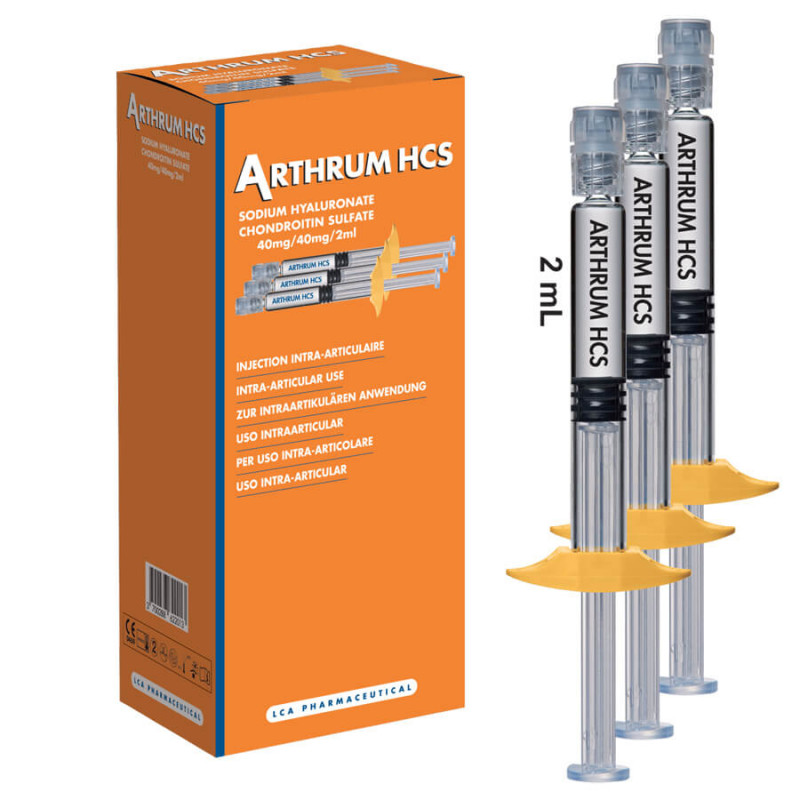
Arthrum HCS 40mg/2ml is indicated for all types of painful synovial joint arthritis, particularly gonarthrosis.
Arthrum HCS is biologically similar to human synovial fluid and participates in the following functions: Protect the cartilage, Protect the synovial membrane, Reduce pain and improve mobility
ARTHRUM HCS is a sterile, injectable viscoelastic device containing two components:
- Sodium Hyaluronate is a glycosaminoglycan derived naturally from hyaluronic acid, obtained by bio-fermentation, with a high molecular weight of 2.8 million Daltons and high concentration (20mg/ml). Sodium Hyaluronate is naturally present in the extracellular matrix, in the soft connective tissues and in the synovial fluid where it is synthesized by the synoviocytes. Being viscoelastic, synovial fluid acts as a lubricant at low frequency, and as a shock absorber at high frequency, limiting mechanical stress and pain in the joint.
- Chondroitin Sulfate is a highly purified, high concentration 20mg/ml glycosaminoglycan of marine origin. Chondroitin Sulfate, naturally present in the connective tissue, is one of the major constituents of the cartilage matrix. Its function is to maintain osmotic pressure by absorbing water and contributing to the hydration of cartilage.
The components of ARTHRUM HCS are not chemically modified, thus eliminating any risk of allergy and potential cytotoxicity and guaranteeing safety and perfect tolerance.
ARTHRUM HCS is a sterile, transparent, homogeneous viscoelastic preparation made from highly purified injectable grades of Sodium Hyaluronate and Chondroitin Sulfate, in accordance with the monographs of the European Pharmacopoeia.
ARTHRUM HCS is a sterile solution with physiological pH, presented in a single-use glass syringe, equipped with a Luer Lock and pre-filled to 2 ml.
The syringe is labelled with the product designation. The syringe is contained in an individual sterility protector.
The syringe, the sterile intra-articular injection needle and the accessories (information leaflet, traceability labels) are included in a packaging case with all the identification details of the ARTHRUM HCS viscoelastic injection device.
ARTHRUM HCS is available in boxes of 1 and 3 syringes.
The pre-filled syringe of ARTHRUM HCS is sterilized by steam pressure in its individual sterility protector. The contents and exterior of the syringe are sterile, so that ARTHRUM HCS can be safely used in the operating room.
The contents of each syringe are sterile and non-pyrogenic.
Sodium Hyaluronate is a glycosaminoglycan naturally present in humans and is a major constituent of the inter-cellular spaces. In the joints, it is a structural component of the cartilage and synovial fluid, which acts as a lubricant, shock absorber, filter, and metabolic intermediate.
Chondroitin Sulfate is also a glycosaminoglycan, an essential component of the cartilage matrix, to which it provides excellent resistance to compression.
ARTHRUM HCS is biologically similar to human synovial fluid and participates in the following functions
Protecting cartilage
- The lubricating properties of the Sodium Hyaluronate and Chondroitin Sulfate molecules contained in ARTHRUM HCS allow the joint surfaces to slide easily over each other, and protect them from mechanical damage.
- The elastic properties of the sodium hyaluronate molecules reduce stress under load, thus protecting the cartilage from excessive compressive forces.
- The high adhesion and covering capacity of Chondroitin Sulfate improves the protection of cartilage and other endo-articular tissues.
Protecting the synovial membrane
The normal synovial membrane lines the joint cavities. It ensures the trophicity of the articular cartilage and the secretion of synovial fluid. It constitutes a filtration and defense barrier.
Sodium Hyaluronate and Chondroitin Sulfate provide a protective barrier by masking the pain receptors present in the synovial membrane.
Reduce pain and improve mobility
- Sodium Hyaluronate and Chondroitin Sulfate replace and improve the viscoelasticity of pathological synovial fluid in osteoarthritic joints.
As a result, they help maintain intra-articular spaces, reduce pain and restore joint mobility.
The therapeutic indications are all types of painful osteoarthritis of the synovial joints, in particular gonarthrosis.
- Check the integrity of the individual sterility protector and the expiration date.
- Aseptically open the individual sterility protector.
- Aseptically pick up the syringe and injection needle.
- Carefully unscrew the stopper from the syringe tip.
- Screw the injection needle onto the Luer Lock system without touching the tip of the syringe.
- Proceed with the intra-articular injection.
- The syringe and needle should be disposed of immediately after use in a dedicated single-use waste container.
Dosage :
Inject ARTHRUM HCS into the affected joint once a week for 3 weeks.
PRECAUTIONS FOR USE:
The following precautions for use are recommended:
- Do not use if the individual sterility protector of the product is damaged.
- Respect the rules of asepsis.
- Do not inject other products at the same time as ARTHRUM HCS.
- Systematically aspirate any synovial fluid effusion beforehand to ensure intra-articular position.
- Precautions for use are those required by the protocol used for intra-articular injections in rheumatology. The specialist physician remains responsible for his or her own techniques and indications.
- It is recommended to inform patients, as for any intra-articular injection, to remain at rest for 24 hours and to avoid any sports or professional activity.
Warnings: The syringe should be used only once, the contents of the syringe should be used immediately after the individual safety shield is opened. Destroy the syringe if the product has not been used and the individual sterility protector has been opened.
Do not resterilize the product in the syringe by heat or any other process.
The risks are loss of physicochemical properties and microbiological contamination of the product, leading to a serious risk of infection and inflammation of the joint.
Do not inject intravascularly.
Do not inject outside the joint cavity or into the synovial tissue or capsule and or in the presence of
presence of a large effusion.
ARTHRUM HCS has not been studied in pregnant women.
It has not been established that ARTHRUM HCS can regenerate cartilage.
Incompatibilities :
Do not use quaternary ammonium (Benzalkonium Chloride) for skin disinfection when using ARTHRUM HCS.
Contra-indications:
- All inflammatory conditions of the joint that should be treated prior to treatment with intra-articular injection of ARTHRUM HCS.
- Do not administer if the patient has a known hypersensitivity to Sodium Hyaluronate and Chondroitin Sulfate.
- ARTHRUM HCS has not been tested in pregnant or nursing women, nor in children under 18 years of age.
Adverse reactions:
ARTHRUM HCS is well tolerated in men. Sometimes during 48 hours a slight pain and or a moderate edema can occur. It is recommended to apply an ice bladder for a few hours.
Composition:
One syringe (2ml) contains 40 mg of sodium Hyaluronate and
40 mg of Chondroitin Sulfate.
Conservation
Protect from light and frost. Store between +2°C and +25°C.
Sinovial® is indicated in cases of pain or reduced mobility in order to restore the physiological and rheological properties of arthritic joints.
Indications: STRUCTUM is used in adults (over 15 years of age) for the treatment of pain and functional discomfort in osteoarthritis of the knee and hip. This drug has a slow onset of action, delayed by about 2 months, and its effect may persist after treatment has been stopped.
The Alvita heating patch offers long-lasting relief from minor aches and pains, joint stiffness and muscle and joint pain.
Box of 4 patches, 13 cm x 9.5 cm
Long-lasting, targeted pain relief. Relieves pain for 8 hours.
Description : This medication is recommended for painful osteoarthritis of the knee, with effusion.
Indications: Piascledine® 300 is a drug indicated for symptomatic treatment (pain and functional discomfort) with delayed effect of osteoarthritis of the hip and knee
Symptomatic treatment ofosteoarthritis.
Arthrum H viscoelastic devices are recommended to maintain the endo-articular space, reduce pain and restore joint mobility by replacing and supplementing the elastoviscosity of the pathological synovial fluid in osteoarthritic joints.
With its flagship active ingredients (CBD, Turmeric, Harpagophytum, Magnesium) Forté Flex Kiné CBD cream helps relieve and soothe muscle and joint pain.
A cream to soothe and limit joint pain Designed to relieve lower back pain, this warming cream has anti-inflammatory and moisturizing properties. It provides a sensation of warmth that helps relax muscles and joints. Mobility improves day by day with regular application.
OSTENIL® Tendon is a medical device indicated for the treatment of pain and limitation of mobility in the case of tendon disorders.
This medicine is used as a ground modifier especially during infectious and viral conditions, flu-like conditions, inflammatory rheumatic conditions and during arthrosis.
Flector effigel is a medicine containing an anti-inflammatory and an analgesic, it is used in the short term local treatment, in adults and children over 15 years old, in case of benign trauma: sprain, strain, or contusion.
Indications: Phytostandard of harpagophytum and willow Pileje is a food supplement to improve joint comfort.
The extracts of harpagophytum and willow are obtained through the patented Phytostandard® process known for its respect for the integrity of the plant's compounds.
Sinovial® HL is indicated for pain or reduced mobility due to degenerative diseases (osteoarthritis), post-traumatic diseases or tendinopathy linked to joint alterations.
Indicated for the symptomatic treatment of osteoarthritis, particularly of the hip and knee. Chondrosulf contains chondroitin sulfate and is indicated for adults (over 15).
Chondrosulf is available in several dosages:
Chondrolsulf 400 mg box 84 capsules
Chondrolsulf 800 mg box 30 capsules
Medical device used as a temporary substitute for synovial fluid in cases of degenerative joint damage, such as osteoarthritis of the knee and hip, which is responsible for a functional change in synovial fluid, without any active synovitis.
Intra-articular injection
Description : GCA2700 Gel combines 16 targeted active ingredients, including Harpagophytum extract, Helichrysum floral water and Gaultheria essential oil with a soothing and relaxing fragrance.