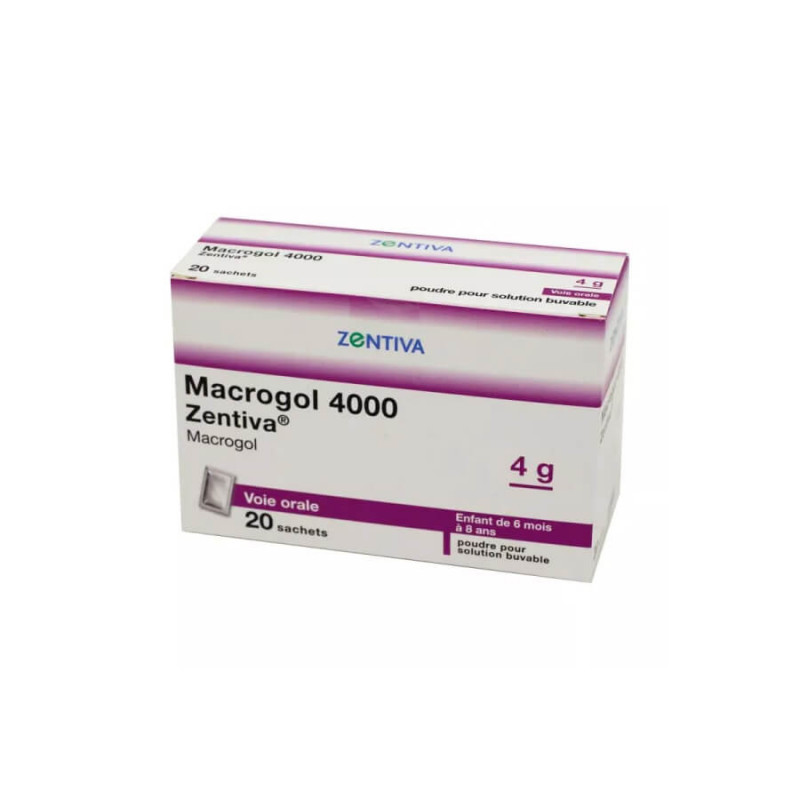
Description ZENTIVA Macrogol 4000 Zentiva Powder Oral Solution 4 g 20 sachets
An organic cause should be ruled out by the physician before initiating treatment, especially in children under 2 years of age. MACROGOL 4000 ZENTIVA should remain a temporary treatment of constipation, for a period not exceeding 3 months, in association with appropriate hygienic and dietary measures. The persistence of the disorders at the end of the treatment, in spite of the maintenance of the hygienic and dietetic measures, will have to make seek and treat an underlying pathology.
Directions for use ZENTIVA Macrogol 4000 Zentiva Powder drinkable solution 4 g 20 sachets
To be taken orally.
Dosage
From 6 months to 1 year: 1 sachet (4 g) per day.
From 1 year to 4 years: 1 to 2 sachets (4 to 8 g) per day.
From 4 years to 8 years: 2 to 4 sachets (8 to 16 g) per day.
The daily dose should be adjusted according to the clinical effects obtained.
The effect of MACROGOL 4000 ZENTIVA is seen within 24 to 48 hours after administration.
Pediatric Population
In children, in the absence of clinical data beyond 3 months, the duration of treatment should not exceed 3 months. The improvement of the intestinal transit induced by the treatment will be maintained by hygienic and dietary measures.
Mode of administration
The content of each sachet must be dissolved in about 50 ml of water just before administration. The treatment should be taken in the morning for a dosage of one sachet per day or divided between morning and evening for a higher dosage. The resulting solution will be clear and transparent as water.
Contraindications:
- Severe inflammatory bowel disease (ulcerative colitis, Crohn's disease) or toxic megacolon,
- Digestive perforation or risk of digestive perforation,
- Ileus or suspicion of intestinal obstruction, symptomatic stenosis,
- Abdominal pain syndromes of undetermined cause,
- Hypersensitivity to the active substance or to any of the excipients mentioned in the section List of excipients.
Special warnings
Efficacy data in children under 2 years of age have been obtained in a limited number of patients.
The medicinal treatment of constipation is only an adjuvant to the hygienic-dietary treatment such as
- enrichment of the diet with vegetable fibers and drinks
- adapted physical activity and re-education of exoneration.
An organic cause must be ruled out before initiating treatment.
After three months of treatment, a clinical assessment of constipation should be performed.
This medicine contains macrogol (polyethylene glycol). Hypersensitivity reactions (anaphylactic shock, angioedema, urticaria, rash, pruritus, erythema) have been reported with macrogol-containing specialties, see Adverse reactions.
In case of diarrhea, patients at risk of fluid and electrolyte imbalance (e.g., elderly, patients with hepatic or renal insufficiency, or those on diuretic therapy) should be monitored and electrolyte control should be considered.
Use with caution in patients with impaired gag reflex and in patients prone to regurgitation or malpractice. Inhalation has been reported with large volumes of polyethylene glycol and electrolytes administered through a nasogastric tube. Children with neurological impairment and oromotor disorders are particularly at risk of inhalation.
Ischemic colitis
Post-marketing surveillance has shown that cases of ischemic colitis, some of them severe, have been reported in patients treated with macrogol as part of a bowel preparation. Macrogol should be used with caution in patients with known risk factors for ischemic colitis or in the concomitant use of stimulant laxatives (such as bisacodyl or sodium picosulfate).
Patients with sudden abdominal pain, rectal bleeding or other symptoms of ischemic colitis should be promptly evaluated.
Special Precautions for Use
Since MACROGOL 4000 ZENTIVA does not contain significant amounts of sugar or polyol, it may be prescribed to patients with diabetes or on a galactose-free diet.
Pregnancy and Lactation :
Pregnancy
Animal studies have not demonstrated direct or indirect deleterious effects on reproduction (see Preclinical Safety Data). There are limited data (less than 300 pregnancies) on the use of MACROGOL 4000 ZENTIVA in pregnant women.
No adverse effects during pregnancy are expected as systemic exposure to MACROGOL 4000 ZENTIVA is negligible. MACROGOL 4000 ZENTIVA can be used during pregnancy.
Nursing mothers
There are no data on the excretion of MACROGOL 4000 ZENTIVA into breast milk.
No effect on nursing infants is expected since systemic exposure to MACROGOL 4000 ZENTIVA in the nursing woman is negligible. MACROGOL 4000 ZENTIVA may be used during lactation.
Fertility
Fertility studies have not been conducted with MACROGOL 4000 ZENTIVA. However, since macrogol 4000 is not significantly absorbed, no effect on fertility is expected.
Effects on Ability to Drive and Use Machines :
No studies on the effects of MACROGOL 4000 ZENTIVA on the ability to drive and use machines have been performed.
Composition ZENTIVA Macrogol 4000 Zentiva Powder for oral solution 4 g 20 sachets
powder for drinkable solution (white to whitish): Macrogol 4000 4 g, qsp 1 sachet.
 Français
Français English
English