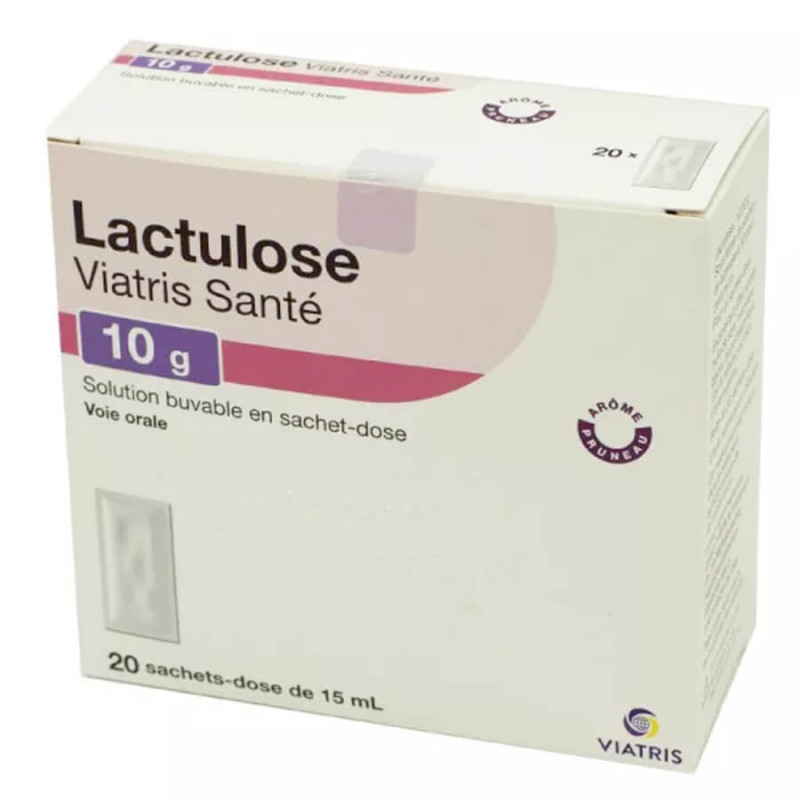
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml is indicated:
- adults, adolescents and children aged 7 to 14 for the symptomatic treatment of constipation.
- in adults for the treatment ofhepatic encephalopathy.
Dosage Lactulose Viatris Santé Oral solution 10 g 20 sachets-dose 15 ml
Dosage should be adapted to individual patient needs.
In the case of a single daily dose, it should be taken at the same time, e.g. with breakfast.
When taking laxatives, it is advisable to drink sufficient fluids (1.5 to 2 liters, i.e. 6 to 8 glasses) during the day.
Constipation
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml can be administered in one or two doses a day.
After a few days, depending on response to treatment, the loading dose should be adjusted to reach the maintenance dose. It may take several days (2 to 3 days) for the treatment to take effect.
The average dosage is as follows
Adults and adolescents :
- attack treatment: 1 to 3 sachet doses per day.
- maintenance treatment: 1 to 2 sachets per day.
Pediatric population
Children aged 7 to 14 :
- initial treatment: 1 sachet-dose per day.
If diarrhea occurs, reduce dosage.
For appropriate dosage in infants and children up to 7 years of age, LACTULOSE BIPHAR 66.5%, oral solution should be used.
Hepatic encephalopathy
In all cases, the ideal dose is that which leads to two or three loose stools per day.
The duration of treatment varies according to symptomatology:
- attack treatment with gastric tube or enema in case of coma or pre-coma:
enema with gastric tube: 6 to 10 sachets diluted in water,
retention enema with balloon tube: 20 sachets in 700 ml of lukewarm water for 30 minutes to 1 hour. If necessary, repeat the procedure 12 hours later.
- oral relay treatment: 1 to 2 sachets, 3 times a day.
Pediatric population
Safety and efficacy in children (newborns to 18 years) with hepatic encephalopathy have not been established. No data are available.
Elderly patients and patients with renal or hepatic impairment
In elderly patients and patients with renal or hepatic impairment, no dosage adjustment is necessary, as exposure to lactulose is negligible.
Method of administration
Oral administration.
May be taken undiluted or diluted in a drink.
The corner of the Viatris Santé Lactulose Oral Solution sachet should be torn open and the contents taken immediately.
The dose of lactulose should be swallowed at once and not kept in the mouth for a prolonged period.
Rectal route.
This medicine must be diluted in water.
Contraindications:
- Hypersensitivity to the active substance or to any of the excipients listed under Excipients.
- Occlusive or subocclusive syndrome.
- Gastrointestinal perforation or suspected perforation.
- Active inflammatory bowel disease (e.g. Crohn's disease, ulcerative colitis).
- Abdominal pain syndromes of undetermined cause.
- Due to the presence of soybean oil, this medicine is contraindicated in cases of peanut or soybean allergy.
Warnings and other special precautions:
Prolonged use of this drug without medical supervision is not recommended.
The usual dosage used in the treatment of constipation is not likely to affect diabetic patients: 30 ml of solution provides 116 kJ (28 kcals). Calories are of carbohydrate origin.
The dosage used in patients with hepatic encephalopathy is usually much higher, and may need to be taken into account in diabetic patients.
Drug treatment of constipation is only an adjunct to dietary hygiene:
- enriching the diet with vegetable fibers and beverages,
- advice on physical activity and exoneration rehabilitation.
Chronic use at inappropriate doses, or misuse, can cause diarrhea and upset the fluid and electrolyte balance. This drug should therefore be used with caution in patients prone to hydro-electrolytic disorders (such as patients with impaired renal or hepatic function, or those receiving diuretics).
When administered as a retention enema, due to the strong cathartic effect, fecal incontinence, bed soiling and perianal irritation due to acidic stools can be expected. The patient's hydration status must be carefully monitored.
It should be borne in mind that the normal functioning of the exoneration reflex may be impaired during treatment.
Information on significant synthetic residues :
This medicine contains lactose, galactose and fructose from the synthetic route. Therefore, patients with galactose or fructose intolerance, total lactase deficiency or glucose-galactose malabsorption syndrome (rare hereditary diseases) should not take this medicine.
Lactulose Viatris Santé Oral Solution should be used with caution in patients with lactose intolerance.
This product contains synthetic sulfites.
Excipients
This medicine contains soybean oil. If the patient is allergic to peanuts or soy, do not use this product.
This medicine contains benzyl alcohol, which may cause allergic reactions.
This medicine contains approximately 30 mg propylene glycol per sachet.
Pediatric population
In infants and children, the prescription of laxatives should be exceptional: it should take into account the risk of impairing the normal functioning of the exoneration reflex.
Pregnancy and lactation :
Pregnancy
Since systemic exposure to lactulose is negligible, no effect is expected in pregnant women.
Lactulose Viatris Santé Oral Solution may be used during pregnancy.
Breast-feeding
Systemic exposure to lactulose in breast-feeding women is negligible, so no effect is expected in newborns or nursing infants.
Viatris Santé Lactulose Oral Solution may be used during breast-feeding.
Fertility
No effect is expected as systemic exposure to lactulose is low.
Effects on ability to drive and use machines :
Lactulose Viatris Santé Solution buvable has no or negligible effect on the ability to drive vehicles and use machines.
Composition Lactulose Viatris Santé Oral Solution
drinkable solution: Lactulose 10 g/15 ml.
Excipients : Prune flavor, (Benzaldehyde, Benzyl alcohol, Butyl acetate, Butylhydroxytoluene, Citronellol, Damascenone, Damascone-alpha, Dihydrocoumarin, Gamma dodecalactone, Ethyl acetate, Ethyl butyrate, ethylmaltol, ethylvanillin, Eugenol, Furfural, Geraniol, hexyl acetate, isobutyric acid, linalool, methylcyclopentenolone, nerol, gamma octalactone, sweet orange peel oil, phenylacetic aldehyde, phenylethyl alcohol, propylene glycol, soybean oil, tartaric acid, 2,6 xylenol), qsp 1 sachet-dose.
Manufacturing process residues: Lactose, Galactose, Fructose, Sulfite.
Notable excipients: soybean oil, propylene glycol, benzyl alcohol.
LACTULOSE MYLAN PHARMA contains significant synthetic residues (see Warnings and Precautions for Use).
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml is indicated:
- adults, adolescents and children aged 7 to 14 for the symptomatic treatment of constipation.
- in adults for the treatment ofhepatic encephalopathy.
Dosage Lactulose Viatris Santé Oral solution 10 g 20 sachets-dose 15 ml
Dosage should be adapted to individual patient needs.
In the case of a single daily dose, it should be taken at the same time, e.g. with breakfast.
When taking laxatives, it is advisable to drink sufficient fluids (1.5 to 2 liters, i.e. 6 to 8 glasses) during the day.
Constipation
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml can be administered in one or two doses a day.
After a few days, depending on response to treatment, the loading dose should be adjusted to reach the maintenance dose. It may take several days (2-3 days) for the treatment to take effect.
The average dosage is as follows
Adults and adolescents :
- attack treatment: 1 to 3 sachet doses per day.
- maintenance treatment: 1 to 2 sachets per day.
Pediatric population
Children aged 7 to 14 :
- initial treatment: 1 sachet-dose per day.
If diarrhea occurs, reduce dosage.
For appropriate dosage in infants and children up to 7 years of age, LACTULOSE BIPHAR 66.5%, oral solution should be used.
Hepatic encephalopathy
In all cases, the ideal dose is that which leads to two or three loose stools per day.
The duration of treatment varies according to symptomatology:
- attack treatment with gastric tube or enema in case of coma or pre-coma:
enema with gastric tube: 6 to 10 sachets diluted in water,
retention enema with balloon tube: 20 sachets in 700 ml of lukewarm water for 30 minutes to 1 hour. If necessary, repeat the procedure 12 hours later.
- oral relay treatment: 1 to 2 sachets, 3 times a day.
Pediatric population
Safety and efficacy in children (newborns to 18 years) with hepatic encephalopathy have not been established. No data are available.
Elderly patients and patients with renal or hepatic impairment
In elderly patients and patients with renal or hepatic impairment, no dosage adjustment is necessary, as exposure to lactulose is negligible.
Method of administration
Oral administration.
May be taken undiluted or diluted in a drink.
The corner of the Viatris Santé Lactulose Oral Solution sachet should be torn open and the contents taken immediately.
The dose of lactulose should be swallowed at once and not kept in the mouth for a prolonged period.
Rectal route.
This medicine must be diluted in water.
Contraindications:
- Hypersensitivity to the active substance or to any of the excipients listed under Excipients.
- Occlusive or subocclusive syndrome.
- Gastrointestinal perforation or suspected perforation.
- Active inflammatory bowel disease (e.g. Crohn's disease, ulcerative colitis).
- Abdominal pain syndromes of undetermined cause.
- Due to the presence of soybean oil, this medicine is contraindicated in cases of peanut or soybean allergy.
Warnings and other special precautions:
Prolonged use of this drug without medical supervision is not recommended.
The usual dosage used in the treatment of constipation is not likely to affect diabetic patients: 30 ml of solution provides 116 kJ (28 kcals). Calories are of carbohydrate origin.
The dosage used in patients with hepatic encephalopathy is usually much higher, and may need to be taken into account in diabetic patients.
Drug treatment of constipation is only an adjunct to dietary hygiene:
- enriching the diet with vegetable fibers and beverages,
- advice on physical activity and exoneration rehabilitation.
Chronic use at inappropriate doses, or misuse, can cause diarrhea and upset the fluid and electrolyte balance. This drug should therefore be used with caution in patients prone to hydro-electrolytic disorders (such as patients with impaired renal or hepatic function, or those receiving diuretics).
When administered as a retention enema, due to the strong cathartic effect, fecal incontinence, bed soiling and perianal irritation due to acidic stools can be expected. The patient's hydration status must be carefully monitored.
It should be borne in mind that the normal functioning of the exoneration reflex may be impaired during treatment.
Information on significant synthetic residues :
This medicine contains lactose, galactose and fructose from the synthetic route. Therefore, patients with galactose or fructose intolerance, total lactase deficiency or glucose-galactose malabsorption syndrome (rare hereditary diseases) should not take this medicine.
Lactulose Viatris Santé Oral Solution should be used with caution in patients with lactose intolerance.
This product contains synthetic sulfites.
Excipients
This medicine contains soybean oil. If the patient is allergic to peanuts or soy, do not use this product.
This medicine contains benzyl alcohol, which may cause allergic reactions.
This medicine contains approximately 30 mg propylene glycol per sachet.
Pediatric population
In infants and children, the prescription of laxatives should be exceptional: it should take into account the risk of impairing the normal functioning of the exoneration reflex.
Pregnancy and lactation :
Pregnancy
Since systemic exposure to lactulose is negligible, no effect is expected in pregnant women.
Lactulose Viatris Santé Oral Solution may be used during pregnancy.
Breast-feeding
Systemic exposure to lactulose in breast-feeding women is negligible, so no effect is expected in newborns or nursing infants.
Viatris Santé Lactulose Oral Solution may be used during breast-feeding.
Fertility
No effect is expected as systemic exposure to lactulose is low.
Effects on ability to drive and use machines :
Lactulose Viatris Santé Solution buvable has no or negligible effect on the ability to drive vehicles and use machines.
Composition Lactulose Viatris Santé Oral Solution
drinkable solution: Lactulose 10 g/15 ml.
Excipients : Prune flavor, (Benzaldehyde, Benzyl alcohol, Butyl acetate, Butylhydroxytoluene, Citronellol, Damascenone, Damascone-alpha, Dihydrocoumarin, Gamma dodecalactone, Ethyl acetate, Ethyl butyrate, ethylmaltol, ethylvanillin, Eugenol, Furfural, Geraniol, hexyl acetate, isobutyric acid, linalool, methylcyclopentenolone, nerol, gamma octalactone, sweet orange peel oil, phenylacetic aldehyde, phenylethyl alcohol, propylene glycol, soybean oil, tartaric acid, 2,6 xylenol), qsp 1 sachet-dose.
Manufacturing process residues: Lactose, Galactose, Fructose, Sulfite.
Notable excipients: soybean oil, propylene glycol, benzyl alcohol.
LACTULOSE MYLAN PHARMA contains significant synthetic residues (see Warnings and Precautions for Use).
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml is indicated:
- adults, adolescents and children aged 7 to 14 for the symptomatic treatment of constipation.
- in adults for the treatment ofhepatic encephalopathy.
Dosage Lactulose Viatris Santé Oral solution 10 g 20 sachets-dose 15 ml
Dosage should be adapted to individual patient needs.
In the case of a single daily dose, it should be taken at the same time, e.g. with breakfast.
When taking laxatives, it is advisable to drink sufficient fluids (1.5 to 2 liters, i.e. 6 to 8 glasses) during the day.
Constipation
Lactulose Viatris Santé Solution buvable 10 g 20 sachets-dose 15 ml can be administered in one or two doses a day.
After a few days, depending on response to treatment, the loading dose should be adjusted to reach the maintenance dose. It may take several days (2-3 days) for the treatment to take effect.
The average dosage is as follows
Adults and adolescents :
- attack treatment: 1 to 3 sachet doses per day.
- maintenance treatment: 1 to 2 sachets per day.
Pediatric population
Children aged 7 to 14 :
- initial treatment: 1 sachet-dose per day.
If diarrhea occurs, reduce dosage.
For appropriate dosage in infants and children up to 7 years of age, LACTULOSE BIPHAR 66.5%, oral solution should be used.
Hepatic encephalopathy
In all cases, the ideal dose is that which leads to two or three loose stools per day.
The duration of treatment varies according to symptomatology:
- attack treatment with gastric tube or enema in case of coma or pre-coma:
enema with gastric tube: 6 to 10 sachets diluted in water,
retention enema with balloon tube: 20 sachets in 700 ml of lukewarm water for 30 minutes to 1 hour. If necessary, repeat the procedure 12 hours later.
- oral relay treatment: 1 to 2 sachets, 3 times a day.
Pediatric population
Safety and efficacy in children (newborns to 18 years) with hepatic encephalopathy have not been established. No data are available.
Elderly patients and patients with renal or hepatic impairment
In elderly patients and patients with renal or hepatic impairment, no dosage adjustment is necessary, as exposure to lactulose is negligible.
Method of administration
Oral administration.
May be taken undiluted or diluted in a drink.
The corner of the Viatris Santé Lactulose Oral Solution sachet should be torn open and the contents taken immediately.
The dose of lactulose should be swallowed at once and not kept in the mouth for a prolonged period.
Rectal route.
This medicine must be diluted in water.
Contraindications:
- Hypersensitivity to the active substance or to any of the excipients listed under Excipients.
- Occlusive or subocclusive syndrome.
- Gastrointestinal perforation or suspected perforation.
- Active inflammatory bowel disease (e.g. Crohn's disease, ulcerative colitis).
- Abdominal pain syndromes of undetermined cause.
- Due to the presence of soybean oil, this medicine is contraindicated in cases of peanut or soybean allergy.
Warnings and other special precautions:
Prolonged use of this drug without medical supervision is not recommended.
The usual dosage used in the treatment of constipation is not likely to affect diabetic patients: 30 ml of solution provides 116 kJ (28 kcals). Calories are of carbohydrate origin.
The dosage used in patients with hepatic encephalopathy is usually much higher, and may need to be taken into account in diabetic patients.
Drug treatment of constipation is only an adjunct to dietary hygiene:
- enriching the diet with vegetable fibers and beverages,
- advice on physical activity and exoneration rehabilitation.
Chronic use at inappropriate doses, or misuse, can cause diarrhea and upset the fluid and electrolyte balance. This drug should therefore be used with caution in patients prone to hydro-electrolytic disorders (such as patients with impaired renal or hepatic function, or those receiving diuretics).
When administered as a retention enema, due to the strong cathartic effect, fecal incontinence, bed soiling and perianal irritation due to acidic stools can be expected. The patient's hydration status must be carefully monitored.
It should be borne in mind that the normal functioning of the exoneration reflex may be impaired during treatment.
Information on significant synthetic residues :
This medicine contains lactose, galactose and fructose from the synthetic route. Therefore, patients with galactose or fructose intolerance, total lactase deficiency or glucose-galactose malabsorption syndrome (rare hereditary diseases) should not take this medicine.
Lactulose Viatris Santé Oral Solution should be used with caution in patients with lactose intolerance.
This product contains synthetic sulfites.
Excipients
This medicine contains soybean oil. If the patient is allergic to peanuts or soy, do not use this product.
This medicine contains benzyl alcohol, which may cause allergic reactions.
This medicine contains approximately 30 mg propylene glycol per sachet.
Pediatric population
In infants and children, the prescription of laxatives should be exceptional: it should take into account the risk of impairing the normal functioning of the exoneration reflex.
Pregnancy and lactation :
Pregnancy
Since systemic exposure to lactulose is negligible, no effect is expected in pregnant women.
Lactulose Viatris Santé Oral Solution may be used during pregnancy.
Breast-feeding
Systemic exposure to lactulose in breast-feeding women is negligible, and no effect is expected in newborns or nursing infants.
Viatris Santé Lactulose Oral Solution may be used during breast-feeding.
Fertility
No effect is expected as systemic exposure to lactulose is low.
Effects on ability to drive and use machines :
Lactulose Viatris Santé Solution buvable has no or negligible effect on the ability to drive vehicles and use machines.
Composition Lactulose Viatris Santé Oral Solution
drinkable solution: Lactulose 10 g/15 ml.
Excipients : Prune flavor, (Benzaldehyde, Benzyl alcohol, Butyl acetate, Butylhydroxytoluene, Citronellol, Damascenone, Damascone-alpha, Dihydrocoumarin, Gamma dodecalactone, Ethyl acetate, Ethyl butyrate, ethylmaltol, ethylvanillin, Eugenol, Furfural, Geraniol, hexyl acetate, isobutyric acid, linalool, methylcyclopentenolone, nerol, gamma octalactone, sweet orange peel oil, phenylacetic aldehyde, phenylethyl alcohol, propylene glycol, soybean oil, tartaric acid, 2,6 xylenol), qsp 1 sachet-dose.
Manufacturing process residues: Lactose, Galactose, Fructose, Sulfite.
Notable excipients: soybean oil, propylene glycol, benzyl alcohol.
LACTULOSE MYLAN PHARMA contains significant synthetic residues (see Warnings and Precautions for Use).
 Français
Français English
English