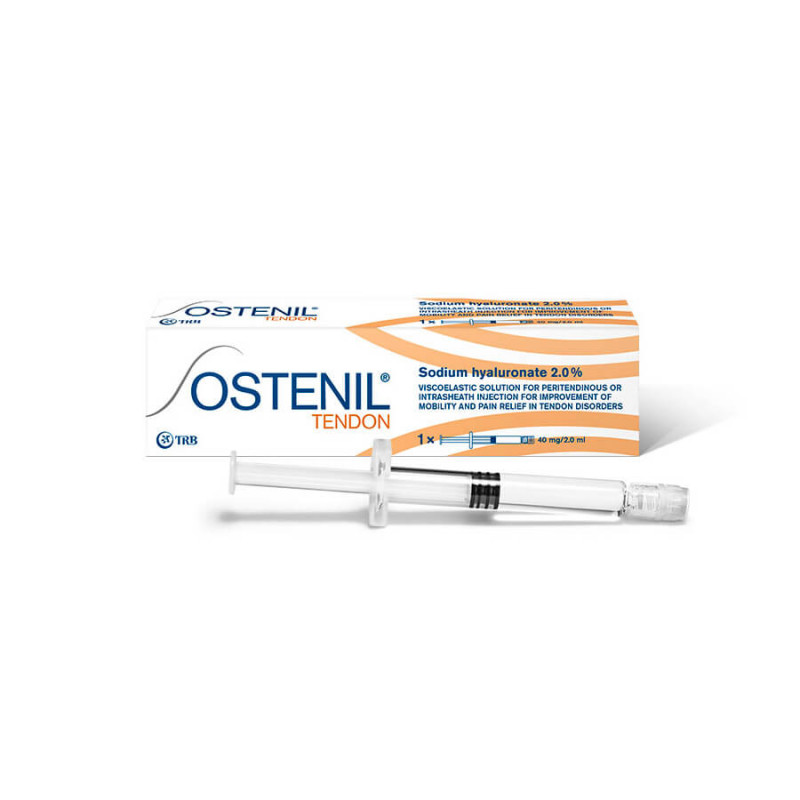
Description OSTENIL® Tendon
OSTENIL® Tendon is a medical device indicated for the treatment of pain and limitation of mobility in case of tendon affections.
OSTENIL® Tendon is a viscoelastic solution of sodium hyaluronate (sodium salt of hyaluronic acid) 2% (40mg/2ml) and mannitol 0.5% (10mg/2ml) for intra-articular injection.
Dosage and administration of OSTENIL® Tendon
Inject OSTENIL® TENDON around the affected tendon or into the affected tendon sheath once a week, for a total of 2 injections. Several tendons can be treated at the same time. Repeated treatments are possible if needed.
The contents and outer surface of the pre-filled syringe of OSTENIL® TENDON remain sterile as long as the sterile packaging remains intact. Remove the pre-filled syringe from the sterile package, unscrew the Luer-Lock closure from the syringe, attach a suitable needle (e.g. 25-27 G) and ensure that it is securely attached by twisting it slightly. Before injection, make sure to remove any air bubbles from the syringe.
Composition OSTENIL® Tendon
1 ml of isotonic solution contains 20.0 mg of sodium hyaluronate, as well as sodium chloride, monosodium phosphate, disodium phosphate, mannitol and water for injection.
Contraindications OSTENIL® Tendon:
Do not use OSTENIL® TENDON in case of known hypersensitivity to any of the components.
OSTENIL® Tendon Interactions:
To date, no data are available on the incompatibility of OSTENIL® TENDON with other products for use on tendons. Adverse reactions:
Following the use of OSTENIL® TENDON, local concomitant manifestations such as pain, warmth, hematoma, redness and swelling may occur.
Precautions:
When using OSTENIL® TENDON, special care should be taken in patients with known hypersensitivity to drugs. As with all invasive treatments, infection may occur in very rare cases. Therefore, general precautions related to peritendinous injections and injections into the synovial tendon sheath should be observed. OSTENIL® TENDON should be injected correctly into the synovial tendon sheath or around the injured tendon, if necessary under scopic control. Nerve damage and injections into blood vessels should be avoided. In the absence of clinical data on the use of OSTENIL® TENDON in children, pregnant or nursing women, and in cases of acute trauma, the administration of OSTENIL® TENDON is not recommended in these patients. Do not use if the pre-filled syringe or sterile packaging is damaged. Any unused solution immediately after opening should be discarded. Otherwise, sterility is no longer guaranteed, and this may be associated with a risk of infection. Store at a temperature between 2°C and 25°C. Do not use after the expiry date mentioned on the box. Keep out of reach of children.
Properties and mode of action:
A strong structure made of fibrous connective tissue, a tendon ensures the transmission of effort from the muscles to the bones and resistance to tension during muscle contraction. Tendons can be surrounded by different structures: for example retinacula, tendon synovial sheaths, peritendon, bursa. Excessive solicitation or inappropriate biomechanical stress can cause inflammation and/or degenerative changes in the tendons, leading to pain and loss of function. Lubricating the tendon may reduce pain, improve tendon function and reduce the possibility of adhesions.
Thanks to its lubricating and viscoelastic properties, OSTENIL® TENDON supports the sliding ability of the tendons as well as the physiological regeneration process. Furthermore, due to its macromolecular structure, OSTENIL® TENDON reduces the free passage of cells and molecules that promote inflammation through the tendon synovial sheath.
OSTENIL® TENDON is a transparent solution based on highly purified natural sodium hyaluronate, produced by fermentation and therefore containing no animal protein. OSTENIL® TENDON also contains mannitol, a free radical scavenger, which helps stabilize the sodium hyaluronate chains. Biocompatibility studies have shown OSTENIL® TENDON to be particularly well tolerated.
Presentation:
One OSTENIL® TENDON pre-filled syringe containing 40 mg/2.0 mL in sterile packaging.
 Français
Français English
English