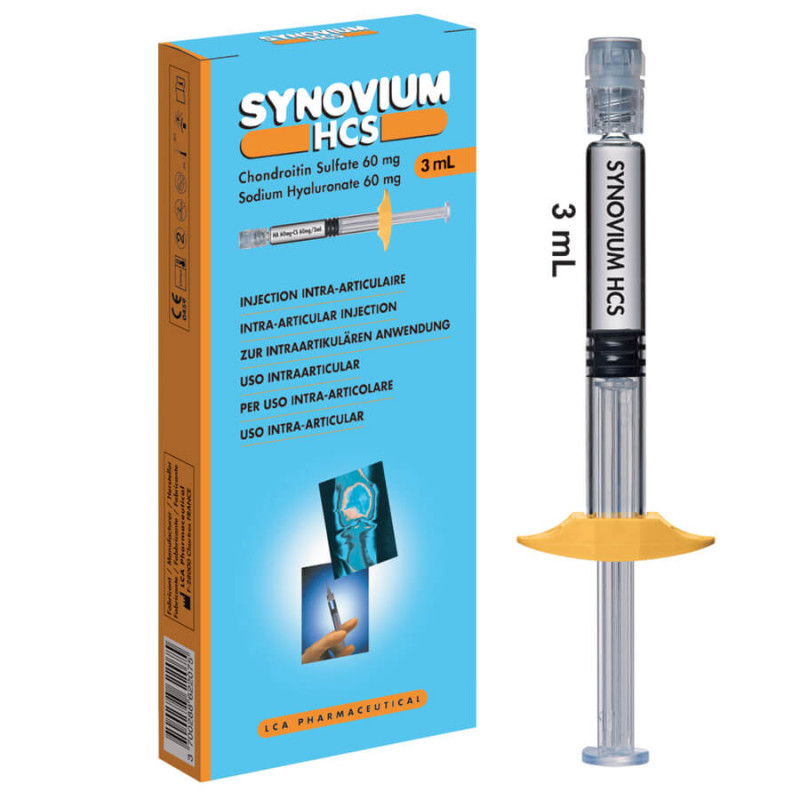
Description Arthrum Synovium HCS 60 mg/60 mg/3 ml arthritis 1 syringe
SYNOVIUM HCS is a sterile viscoelastic device containing two active ingredients:
- A natural derivative of hyaluronic acid, sodium hyaluronate, obtained by bio-fermentation, not chemically modified, high molecular weight ≥ 2,800,000 Daltons and high concentration 20mg/ml (i.e. 60 mg per intra-articular injection). Sodium hyaluronate is naturally present in the synovial fluid, it is synthesized by synoviocytes.
Its mechanical properties change according to movement. Thanks to its viscoelasticity, it plays the role of lubrication at low frequency and the role of shock absorber at high frequency. It has an analgesic action by reducing the sensitivity of mechano-nociceptive ion channels.
- Chondroitin sulfate is a glycosaminoglycan present in connective tissue and a component of the cartilage matrix. Its function is to maintain osmotic pressure by absorbing water and to help hydrate cartilage. More importantly, it serves as a chondroprotective agent by protecting cartilage from enzymatic reactions and free radical damage.
Form and presentation Arthrum Synovium HCS 60 mg/60 mg/3 ml arthritis 1 syringe
SYNOVIUM HCS is a sterile, transparent, homogeneous, chemically unmodified viscoelastic preparation composed of highly purified sodium hyaluronate and Chondroitin Sulfate, European Pharmacopoeia grade for injection. SYNOVIUM HCS is a sterile, isotonic solution, with physiological PH, presented in a sterile, single-use, luer lock syringe, pre-filled with 3 ml in a box of one syringe. The syringe is labelled with the product designation. The syringe is presented in an individual sterility protector. The syringe and the information leaflet are included in a packaging case containing all the identification details of the viscoelastic implantable device:
SYNOVIUM HCS Box of 1 syringe.
Composition Arthrum Synovium HCS 60 mg/60 mg/3 ml arthritis 1 syringe
Each syringe of SYNOVIUM HCS contains :
Sodium hyaluronate (F.B.) 60 mg
Chondroitin Sulfate 60 mg
Sodium chloride 21 mg
Borate buffer pH 7.2 ± 0.2
Water for injection qsp 3.0 ml
The sterilization method is by steam autoclave.
The content of each syringe is sterile and non-pyrogenic.
Properties Arthrum Synovium HCS 60 mg/60 mg/3 ml arthritis 1 syringe
Sodium hyaluronate occurs naturally in humans and is a major constituent of the inter-cellular spaces. In the joint, it is a structural component of the cartilage and synovial fluid which acts as a lubricant, shock absorber, filter and metabolic medium. Chondroitin sulfate is also a glycosaminoglycan, an essential component of the cartilage matrix that provides excellent resistance to compression.
SYNOVIUM HCS is biologically similar to human synovial fluid. Its functions are as follows:
Protect the cartilage
- The lubricating properties of the sodium hyaluronate and chondroitin sulfate molecules contained in SYNOVIUM HCS ensure that the joint surfaces slide perfectly against each other and protect them from mechanical damage.
- The elastic properties of the sodium hyaluronate and chondroitin sulfate molecules reduce the load stress and protect the cartilage from compressive forces.
- The strong covering power of Chondroitin Sulfate allows to improve the protection of the cartilage (chondro-protection) and the other endo-articular tissues.
Cartilage nutrition
- Small molecules such as water, electrolytes and nutrients can easily diffuse to the cartilage and the synovial membrane.
Protecting the synovial membrane
- Sodium hyaluronate provides a protective barrier by masking the pain receptors of the synovial membrane.
Sodium hyaluronate is normally present in the synovial fluid of the healthy joint, and is deteriorated in both concentration and molecular weight in the osteoarthritis joint. In the treatment of osteoarthritis, SYNOVIUM HCS therefore has the effect of improving the deficient rheological characteristics of the synovial fluid (viscosity and elasticity). These characteristics are important for the absorption of shocks as well as for the lubrication and protection of cartilage surfaces.
Indications Arthrum Synovium HCS 60 mg/60 mg/3 ml
SYNOVIUM HCS viscoelastic devices are indicated for the symptomatic treatment of gonarthrosis, in particular to reduce pain and restore joint mobility by replacing and supplementing the elasto-viscosity of the synovial fluid in arthritic joints.
The therapeutic indications are all types of painful gonarthrosis:
- Primary gonarthrosis (Kelgreen radiological stages I, II and III)
- Gonarthrosis and associated general factors:
- Ineffectiveness of usual treatments.
- Intolerance and/or contraindication to NSAIDs and/or analgesics.
- Taking anticoagulants, poly-medication (hypertension, diabetes, obesity, cardiovascular and gastrointestinal problems).
- Contraindications related to the fitting of a prosthesis: young patients and various contraindications related to the patient's condition.
- Early gonarthrosis in young patients.
- Gonarthrosis secondary to trauma and the aftermath of joint fractures.
MODE OF ADMINISTRATION :
SYNOVIUM HCS should be injected intra-articularly by a physician.
- Check the integrity of the individual sterility protector and the expiration date.
- Open the individual sterility shield aseptically.
- Aseptically pick up the syringe.
- Screw the injection needle onto the luer lock after removing the tip-cap without touching the tip of the syringe with your finger
of the syringe with your finger.
- Proceed with the intra-articular injection.
The contents of the syringe are for single use only.
- The syringe and needle must be discarded immediately after use.
They will be disposed of in a specific single-use waste box.
DOSAGE:
SYNOVIUM HCS is for adult use only.
The dosage schedule for SYNOVIUM HCS is one intra-articular injection into the knee.
A second injection may be repeated between the first and third month if the patient's pain symptomatology warrants it.
PRECAUTIONS FOR USE :
The following precautions for use are recommended:
- Check the integrity of the individual sterility protector of the product before use.
- Respect the rules of asepsis.
- Do not inject other products at the same time as SYNOVIUM HCS.
- Precautions for use are those required by the protocol used for intra-articular injections in rheumatology and orthopedic surgery.
The specialist physician remains responsible for his or her own techniques and indications.
- It is recommended to inform patients, as for any intra-articular injection, to remain at rest for 24 hours and to avoid any sports and/or professional activity.
Warnings:
Do not inject intravascularly.
Do not inject outside the joint cavity or into the synovial tissue or capsule and or in the presence of a significant
presence of a large effusion.
SYNOVIUM HCS has not been studied in pregnant women.
INCOMPATIBILITIES :
Do not use quaternary ammonium (Benzalkonium Chloride) for skin disinfection
during intra-articular injection of SYNOVIUM HCS.
CONTRAINDICATIONS :
- All inflammatory pathologies of the joint that should be treated before undertaking
treatment with SYNOVIUM HCS intra-articular injection.
- Do not use if the patient has a known hypersensitivity to sodium hyaluronate.
ADVERSE REACTIONS:
SYNOVIUM HCS is well tolerated in humans. Occasionally, pain may occur during 48 hours.
It is recommended to apply an ice bladder for a few hours.
STORAGE:
Protect from light and freezing. Store between +2°C and +25°C.
After opening, SYNOVIUM HCS viscoelastic device must be used immediately and discarded after use
after use.
 Français
Français English
English